What’s new in biology: May 2026
A cure for congenital deafness, recreating snake venom, antibodies, a legend in cardiovascular medicine, and a successful hair loss treatment?
Niko McCarty and Saloni Dattani review important things happening in the world of biotechnology and medicine.
This is the last week to apply to Invisible College, our week-long seminar in Cambridge in August. If you are 18–22 years old, bright, ambitious, and interested in ideas: apply! Applications close on Friday 8th May.
The first gene therapy for deafness earns FDA approval. About sixty percent of all babies born deaf have some underlying genetic cause, and mutations in the OTOF gene account for between 2 and 8 percent of them. That gene encodes a protein called otoferlin, which enables hair cells inside the ear to transmit messages into the brain.
The OTOF gene is quite long, though, stretching about 90,000 bases of DNA. This means it’s too long to ‘package’ inside a single viral capsid for delivery. Regeneron, the company that developed the therapy, therefore decided to split up the gene, package each piece separately, and then deliver each half in its own viral capsid. The therapy was injected directly into the fluid-filled space of the inner ear, called the perilymph.
In one study with 12 children – all of whom were unable ‘to hear a gas-powered lawn mower’ before starting therapy – six could hear soft whispering without any hearing aids after 24 weeks of the therapy, and three others developed normal hearing. Besides being the first gene therapy for deafness, this is ‘also the first dual-AAV [adeno-associated viral capsid] therapy to be approved by the FDA’, writes Veera Rajagopal. The drug will be given away for free to patients in the United States, according to a Regeneron press release.
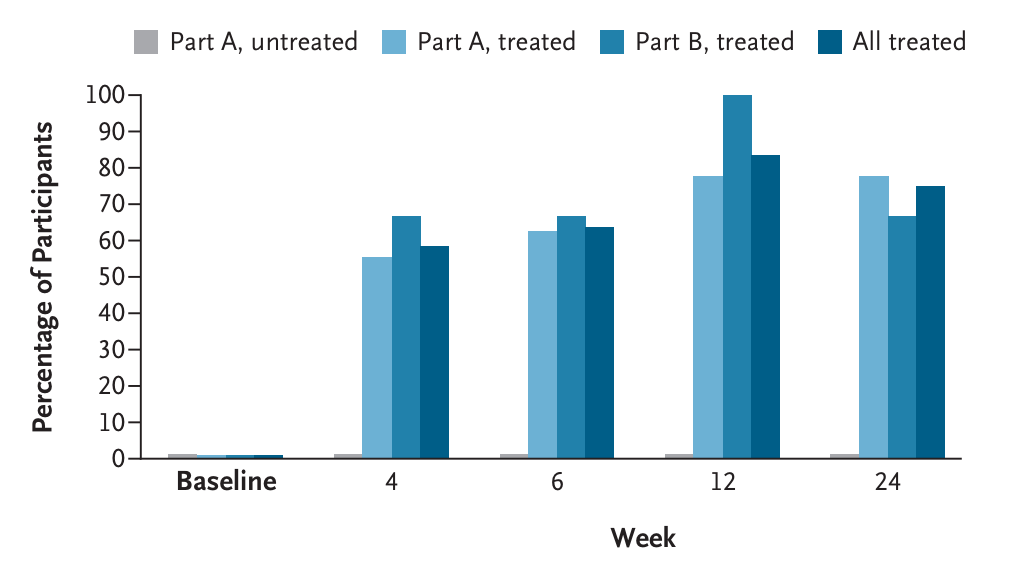
Does hair loss treatment actually work now? A new oral drug for hair loss actually seems to work, which feels surprising in a field that’s been plagued by scams and overblown claims for decades. Until now, there have been only a few approved treatments: oral finasteride (only for men, but coming with side effects like sexual dysfunction) and topical minoxidil (for men and women, applied to the scalp, with effects that are not long-lasting).
Now, Veradermics reports their drug, VDPHL01, grew around 30 additional hairs per square centimeter over six months compared to roughly 7 in the placebo group in a late-stage trial, and around 80 percent of participants reported improvement. The drug is a reformulated version of minoxidil engineered for slower, steadier release: standard oral minoxidil has a half-life of only a few hours; the idea now is to keep levels consistently high enough for continuous regrowth without reaching toxic levels. A second confirmatory trial in men is expected to report by the end of the year, which could result in FDA approval if it’s successful. There’s also an ongoing trial in women, where the options are even more limited.
Scientists watched a protein fold in real-time. A protein moves between its folded and unfolded forms in less than one-millionth of a second. For a new study, researchers captured this transition in real time for eight small proteins. They measured each protein’s folding by attaching red and green fluorescent dyes to amino acids that sit far apart when unfolded but come together when folded. As the dyes get closer, the green dye transfers energy to the red dye instead of emitting its own light. Whereas an unfolded protein will emit roughly equal numbers of green and red photons, a folded protein will emit mostly red photons. After watching these proteins fold, using high-powered microscopes, the scientists discovered (surprisingly) that large proteins fold faster than small ones. The smaller proteins had shorter waiting times – meaning they flip back and forth between states more frequently – but larger proteins had shorter transition times, meaning they completed folds faster once the process had begun. The largest protein transitioned in 0.7 microseconds, compared to 3.1 for the smallest. Evolution has probably optimized larger proteins to fold more efficiently via cooperativity, where one part of the protein coaxes another part to snap into place.
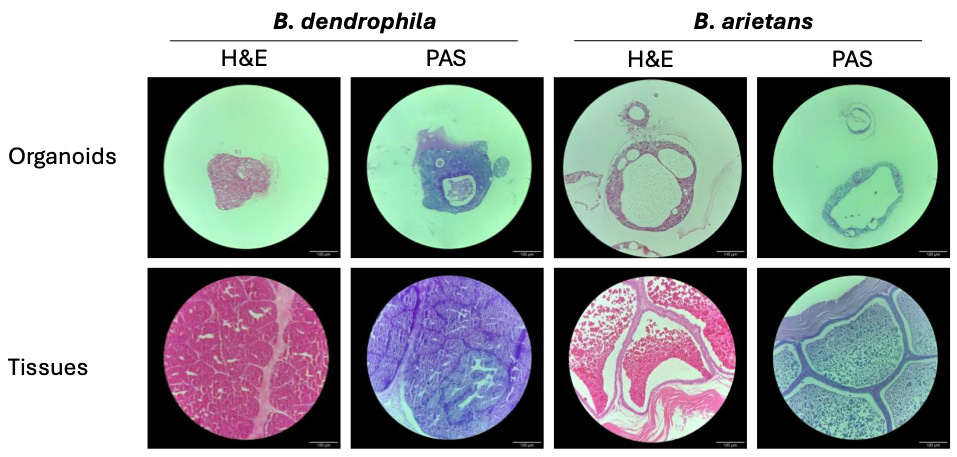
Researchers recreate snake venom with organoids, to help research into anti-venoms. Snakebites kill 50,000 people each year in India alone. Computational biologists are using AI tools, such as RFdiffusion, to design proteins that can neutralize a few toxins in snake venom, and which don’t need refrigeration, but so far they only work against select proteins in the venom rather than the whole cocktail. To make antivenom today, scientists physically milk a snake by pressing on its venom glands so that it bites down and releases venom into a container. Then, they inject small, non-lethal doses of that venom into a horse, whose immune system produces the antibodies that are turned into the final antivenom. There are many ways to make this process far better!
In 2020, for example, Dutch researchers made organoids – spherical clusters of tissue grown from stem cells – for front-fanged snakes, including Elapidae and vipers. The nice part about these organoids is that they actually make venom that is chemically identical to the venoms made by living snakes. But for several years, nobody was able to make organoids for non-front-fanged snakes, which constitute about 70 percent of all snake species on Earth. These snakes have a different gland anatomy — they produce venom in a place called Duvernoy’s gland, which is smaller and harder to access than the venom glands of vipers and elapids, and extracting stem cells from it requires anesthetizing the snakes first. Recently, though, English scientists figured it out.
A new preprint describes how to make venom-producing organoids for snakes from the Colubridae family, which is the largest snake family on Earth and includes king, rat, and garter snakes. These organoids only make a tiny amount of venom (you’d need hundreds or thousands of them just to make enough to inject a single horse), but will be a useful starting point for basic research into antivenom design.
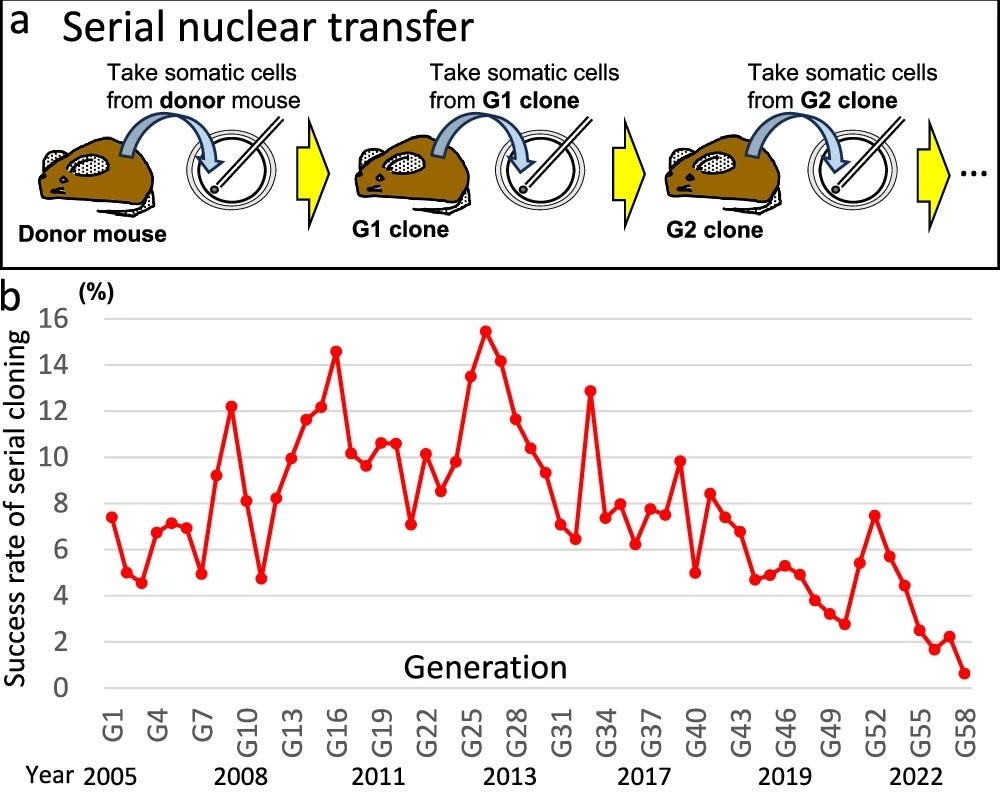
How long can you clone mice for? Scientists cloned a mouse for the first time in 1997 by taking a cell from an adult animal and injecting it into an egg lacking a nucleus. The egg, after being stimulated with chemicals, began to divide and gave rise to a mouse which was a genetic copy of the original animal.
Now the same research group have published a paper showing how they kept cloning the mice, repeatedly, for a period of 20 years. One might suspect that a lack of genetic diversity would make the animals non-viable (after all, that’s what happened to royal dynasties in which brothers married sisters or cousins). But instead, the researchers found that the cloning success rate stayed basically flat through 40 generations, but then started to fall.
The experiment ended after 58 generations, when the scientists could no longer clone the mouse. ‘By generation 57, the cloned mice had acquired over 3400 single-base changes relative to the starting mouse, whereas 62 generations of natural reproduction (in an inbred mouse strain) had accumulated 752’, writes Merrick Pierson Smela, a geneticist. ‘In other words, cloning caused 3.1 times more single-nucleotide mutations per generation than natural reproduction’. The mice also lost an entire X chromosome between generation 25 and 45, which was never regained.
Some ask ‘Where’s my flying car?’ but I ask ‘Where’s my omnibus p53 cancer drug?’ p53 is the body’s most important protein for protecting us against cancer. Roughly half of all solid cancers develop mutations in it that prevent tumor cell death, promote tumor growth, and prevent DNA damage repair. So why don’t we have effective drugs against it?
Unfortunately, there are thousands of different p53 mutations, and most work by changing the protein’s shape rather than knocking out a specific function, which makes it hard to fix with a drug. What’s worse is that mutant p53 is harder for cells to break down, so it builds up in high levels in cancer cells; drugs have a lot of dysfunctional protein to overcome.
Progress in p53 drug development seems to have been pretty hard. But some mutants are easier to target than others – like the Y220C mutant of p53, which is shaped with a pocket that a small molecule can slot into. That’s what some researchers have been targeting. A new phase 1 trial shows a proof-of-concept that this strategy can work using rezatapopt, a new compound that binds to the Y220C mutant p53 and reduces its levels in patients’ blood.
Although itʼs promising, this mutation is only carried by around 1 percent of p53 mutant tumors! And there’s still more to do to demonstrate its efficacy; various previous attempts at p53 drugs have failed in late-stage clinical trials. It’s a difficult problem, but one that would be hugely impactful if solved. Let’s hope this is a successful step towards it.
Roughly a third of approved antibody drugs bind to proteins they’re not meant to. Monoclonal antibodies are used in medical treatment for various conditions including many cancers, and are generally described as extremely precise drugs that lock onto a single target – which is true, relative to other drugs – but it turns out that some also bind to unintended targets.
A new study screened 74 antibodies that were FDA-approved or in clinical studies against over 6,000 human proteins. The authors found that 28 percent bound to at least one unintended target, which could cause unexpected side effects or reduce the efficacy of the drugs, although that wasn’t tested directly. On the bright side, the authors showed that inducing just one or two mutations to the antibody were enough to remove these off-target bindings without touching the intended ones. This kind of early systematic screening isn’t standard practice yet, but it could probably help find better antibody candidates and reduce failure rates in clinical trials.
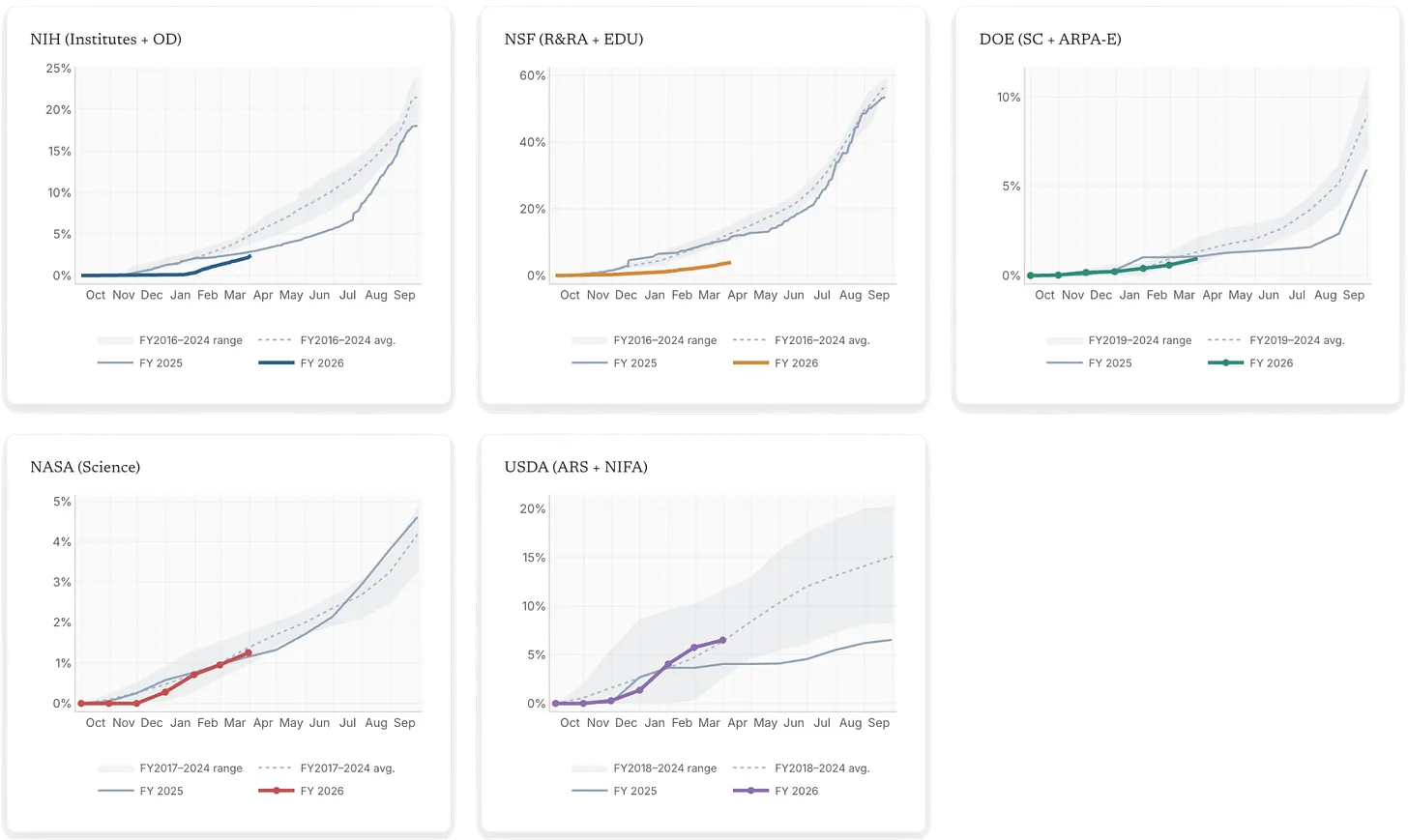
US science agencies have money but aren’t spending it. A new analysis tracking real-time spending data finds that NSF is issuing new grants roughly 70 percent slower than its historical pace, NIH about 50 percent slower than before, and the National Cancer Institute particularly bad at 79 percent slower. While Congress actually held firm on science budgets, the current administration has been slow-walking the actual disbursement of funds to researchers, as it did in FY2025 before advocacy pressure forced a correction.
To make it easier to understand these slowdowns, Jordan Dworkin, Saloni’s colleague at Coefficient Giving, built sciencespending.org, a public tracker of grant-making pace across five major agencies. The problem is worse than just delays, because unspent NIH funds expire at the end of the fiscal year and go back to the Treasury, which means that money may never reach researchers.
Eugene Braunwald, probably the most important figure in modern cardiology, died on April 22nd at age 96. It’s hard to overstate his influence. Before his work in the 1970s, a heart attack was treated with sedation and bed rest: the prevailing view was that once muscle was dying, little could be done. Braunwald overturned this by showing that the amount of damage from a heart attack depended on the balance between oxygen supply and demand in the heart, and that one could act fast to shift this balance and rescue threatened muscle. The idea became known as ‘time is muscle’ and underpins almost all modern reperfusion therapy: clot-busting drugs, angioplasty, and stenting, which help clear arteries and deliver oxygen to the heart quickly.
He also defined the mechanics of hypertrophic cardiomyopathy (a condition where the heart muscle thickens and obstructs blood flow), and in 1984, he founded the TIMI study group, which has since run over 70 randomized trials shaping standard care for heart attacks, heart failure, and diabetes.
Over the course of his career, the share of patients who died within a month after having the most severe type of heart attack fell roughly three-fold – from a 30-day mortality rate of around 17 percent to around 6 percent; much of that is attributable to treatments based on his research.
It’s striking how recent all of this is: the idea that a heart attack was something you could actively treat is only about 50 years old! (Part of the reason is that people hadnʼt invented ways to study the mechanics of the living heart without killing the patient until the mid-20th century.)



statnews links for Veradermics are broken btw